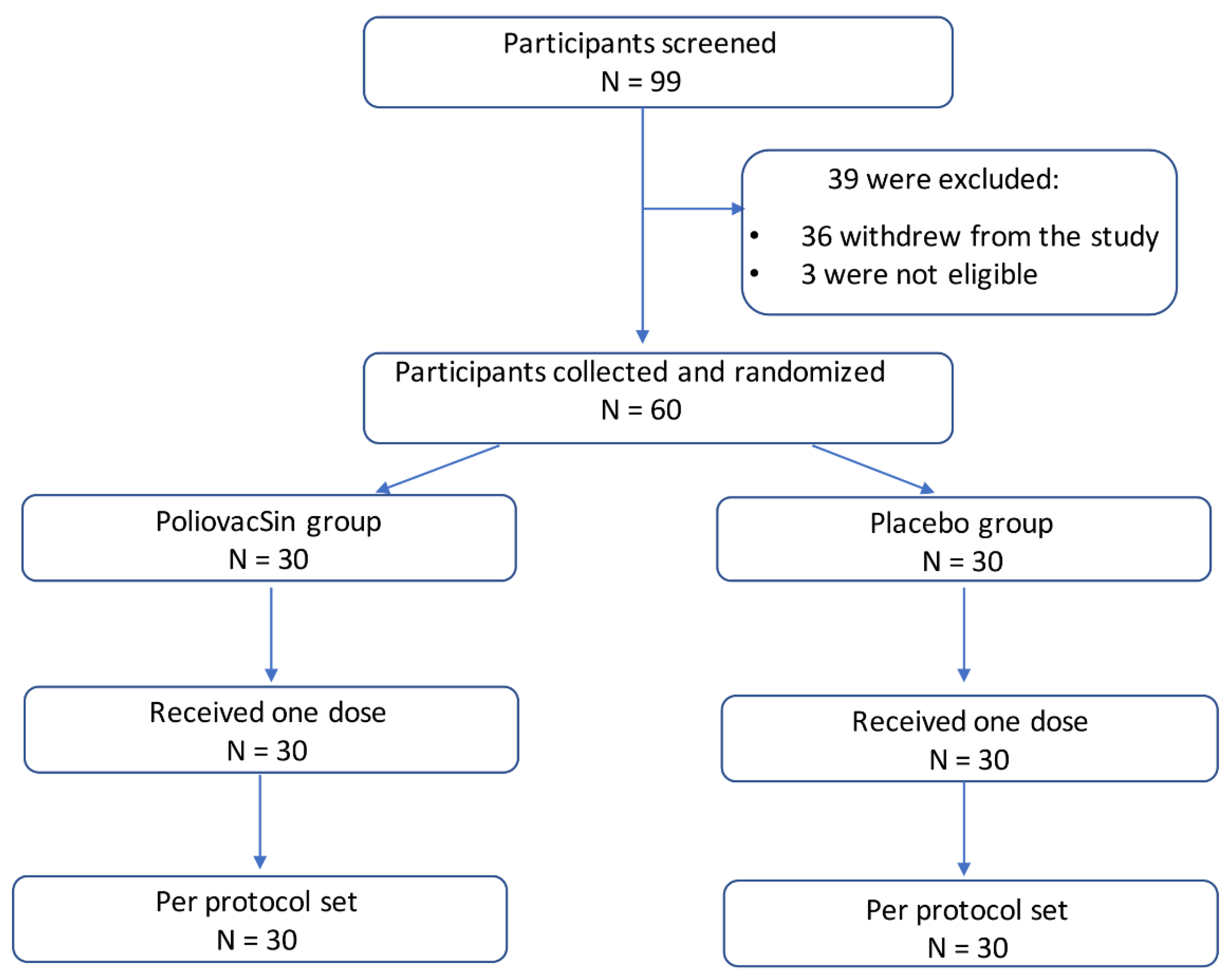
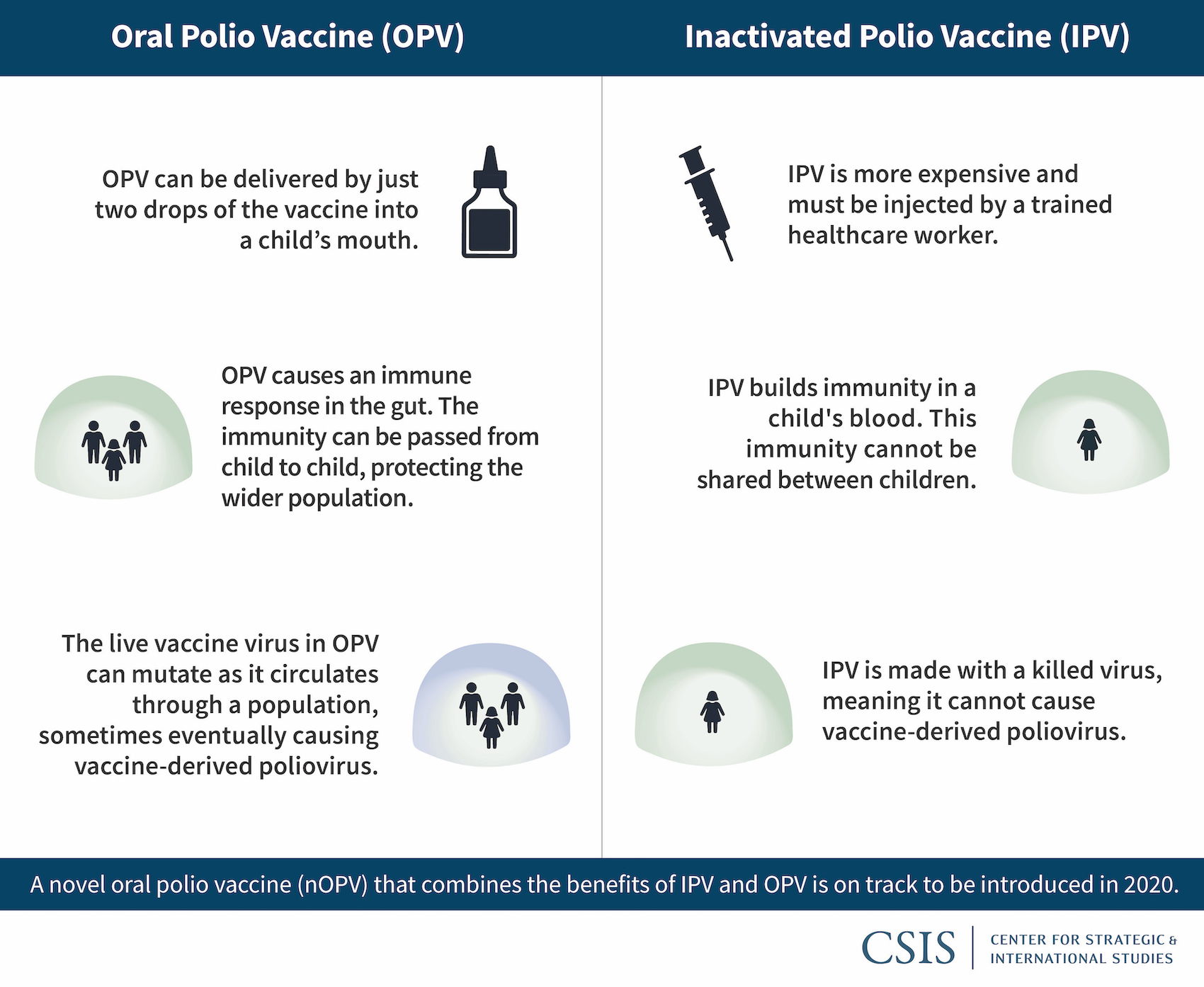
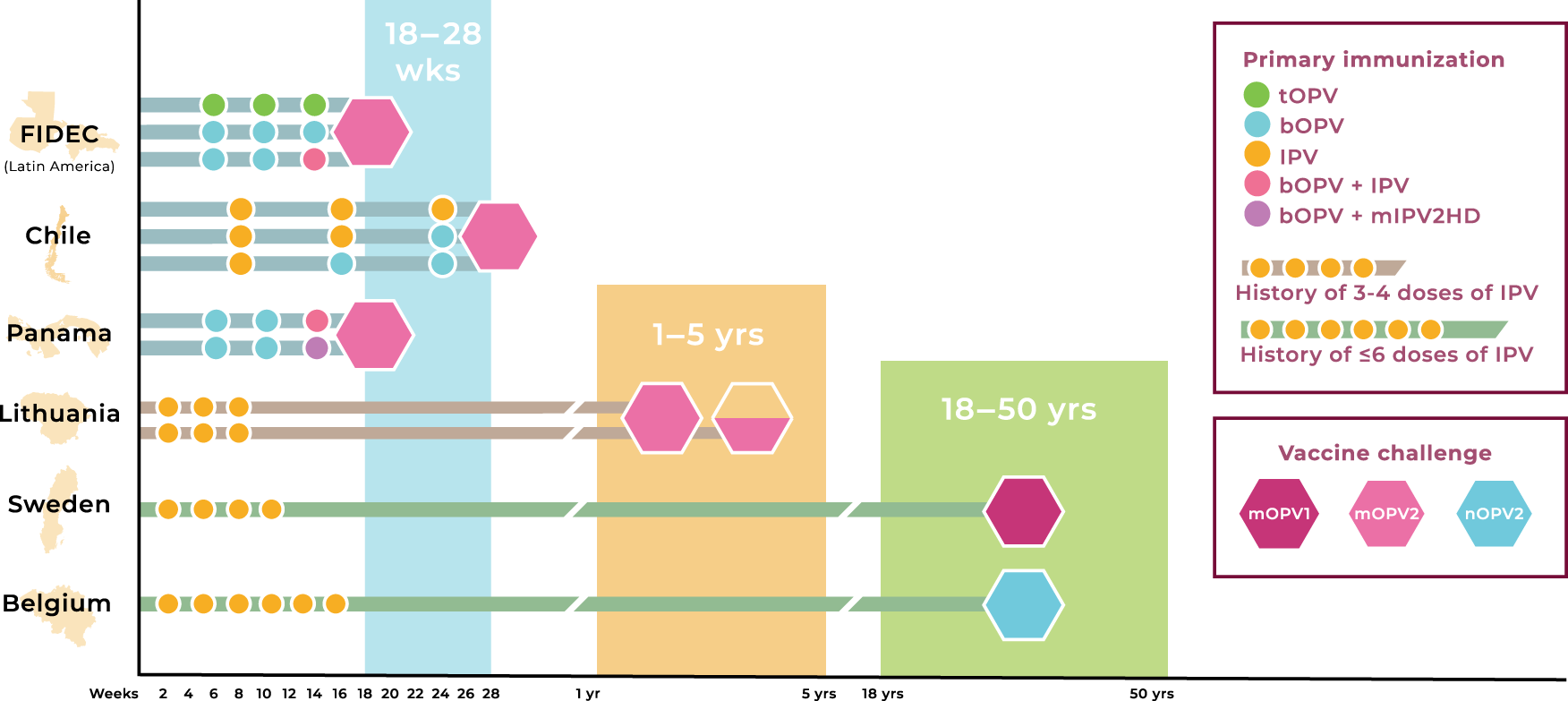
The safety and immunogenicity of two novel live attenuated monovalent (serotype 2) oral poliovirus vaccines in healthy adults: a double-blind, single-centre phase 1 study - The Lancet

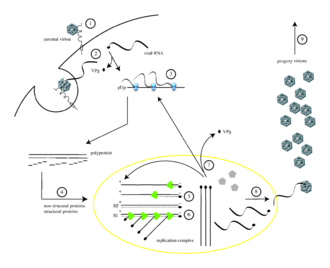
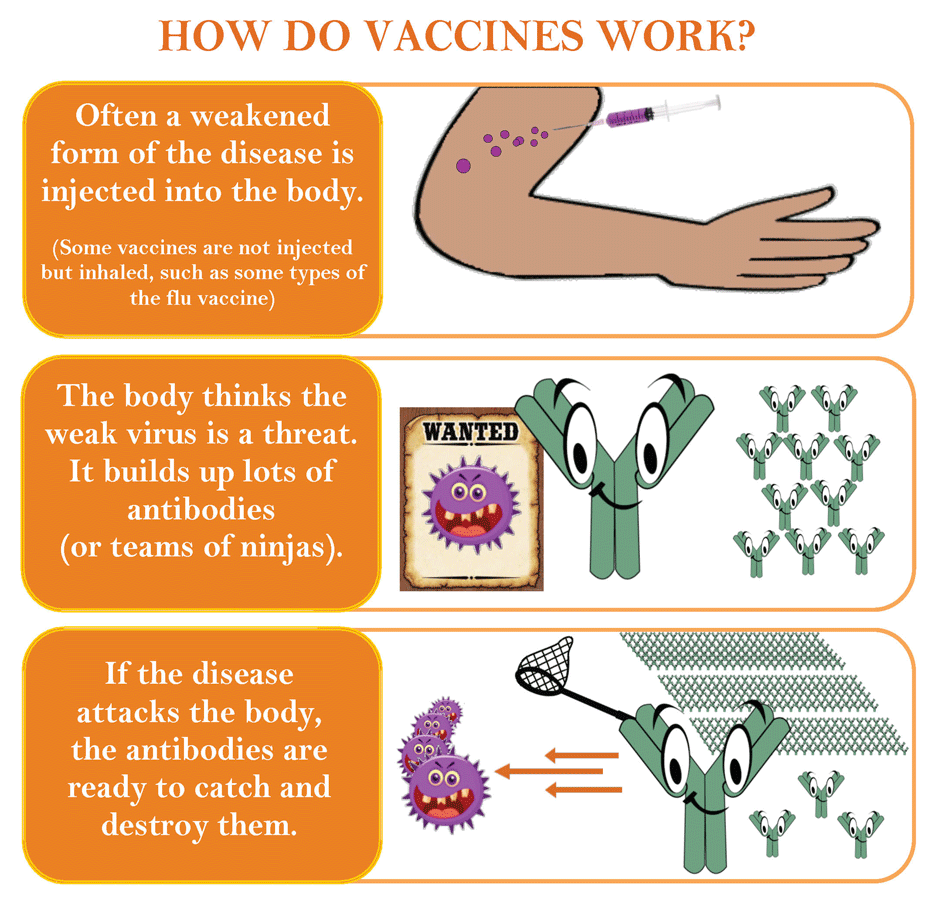
Frontiers | Poliovirus Vaccination Induces a Humoral Immune Response That Cross Reacts With SARS-CoV-2
Next Generation Inactivated Polio Vaccine Manufacturing to Support Post Polio-Eradication Biosafety Goals | PLOS ONE
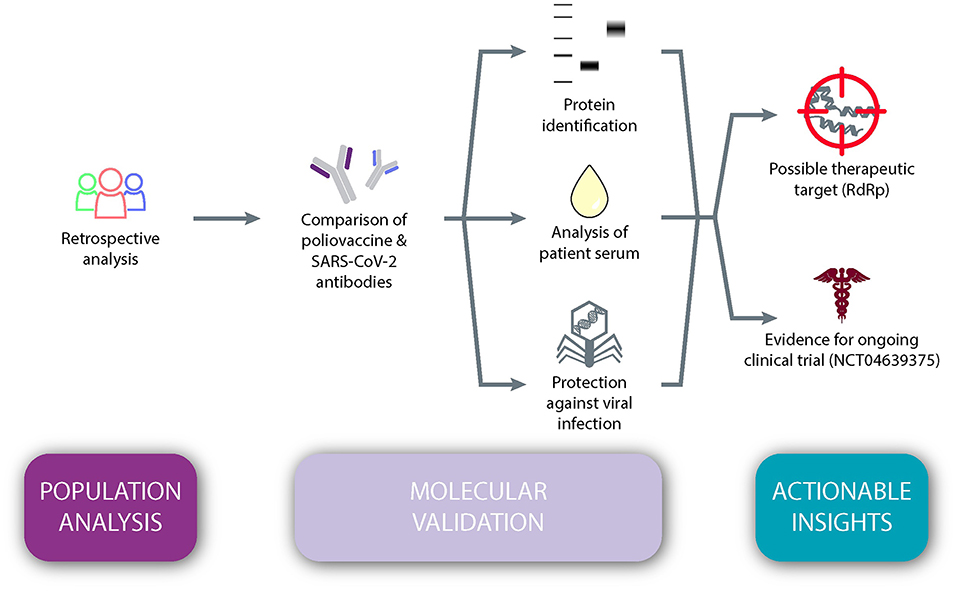
Frontiers | Poliovirus Vaccination Induces a Humoral Immune Response That Cross Reacts With SARS-CoV-2