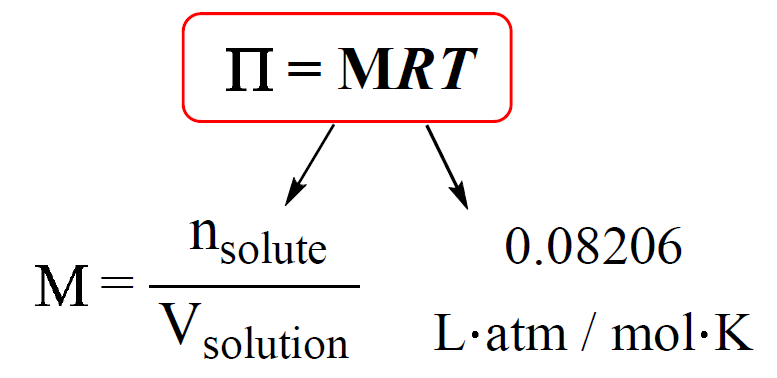
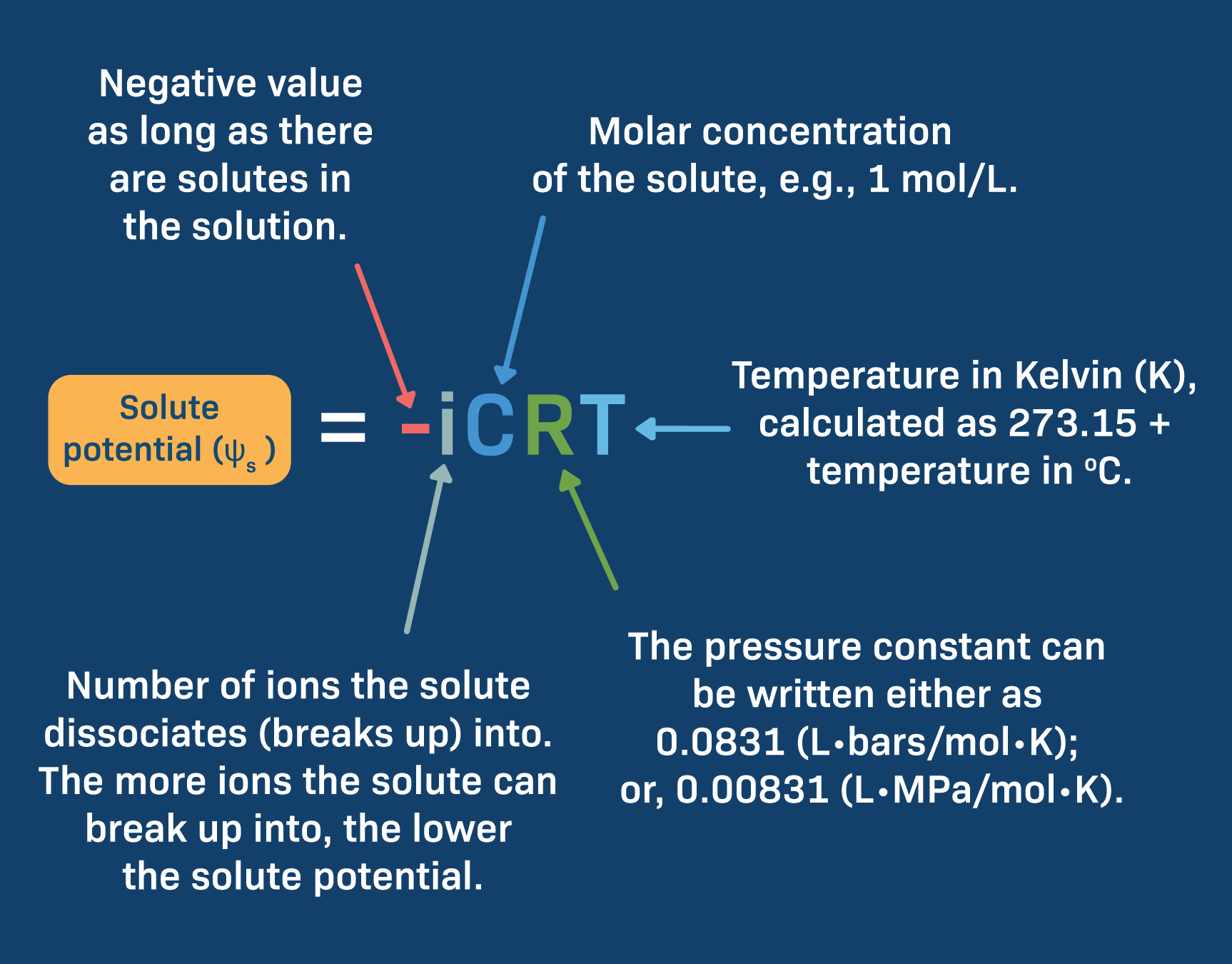
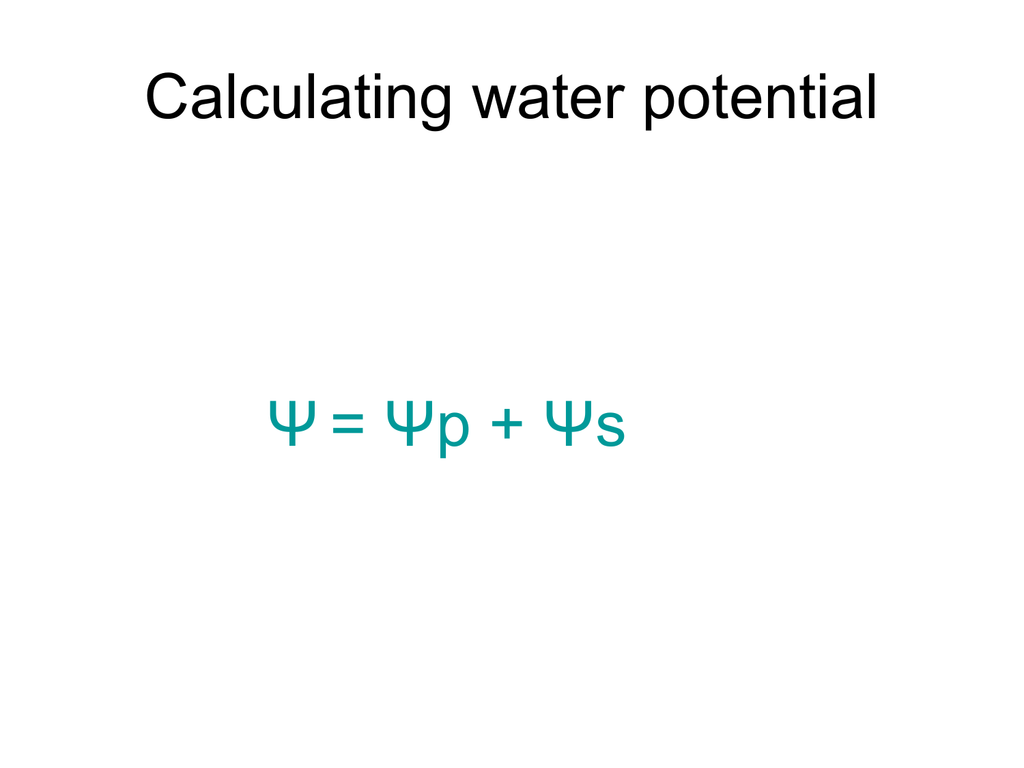SOLVED: Calculate the solute potential of a 2.0 M sucrose solution at 20oC under standard atmospheric condition. Take into account that sucrose doesn't ionize in water, so its ionization constant (i) is

Understanding Water Potential. Water Potential Water potential predicts which way water diffuses through plant tissues and is abbreviated by the Greek. - ppt download

Water Potential Osmosis. Water potential The combined effects of 1.) solute concentration 1.) solute concentration 2.) physical pressure (cell wall) - ppt download

SOLVED: Calculate how increasing the molar concentration of a sucrose solution affects the sucrose solution's solute potential. a) The equation for solute potential is: ψS = −iCRT, where i = the ionization